+86 17737144966
Lithium-ion capacitors (lics) are a new type of asymmetric supercapacitors that combine lithium-ion battery lithium-ion embedded electrodes with the double-layer electrodes of the supercapacitor. Generally, practical lithium-ion capacitors consist of capacitive cathode (active carbon) and battery-type anode (graphite, soft carbon, hard carbon) . It is inferred that in the case of overcharge, the positive potential may exceed the electrolyte oxidation decomposition limit. Therefore, the overcharge behavior of li-ion capacitors mainly depends on the positive electrode of activated carbon. Therefore, it is necessary to study the overcharge behavior of AC cathode in non-aqueous lithium-ion electrolyte without the interference of anode electrode.
Changes in energy efficiency can be used as a useful tool to determine the upper and lower limits of the potential stability of AC electrodes in aqueous or non-aqueous electrolytes. According to the charge-discharge curve of activated carbon half-cell, EE is defined as the ratio of discharge energy to charge energy. In addition, Coulomb efficiency (CE) is called the ratio of discharge capacity to charge capacity, and voltage efficiency (VE) is called the ratio between discharge platform voltage and charge platform voltage of activated carbon. When a delicate redox (charge transfer reaction) occurs at the electrode-electrolyte interface on the AC electrode, the electrolyte decomposition reaction results in the deposition of insoluble products and the passivation film (cathode electrolyte interface) on the surface of the AC electrode or in the contact pores. Here, EE is more sensitive to irreversible redox than CE due to the cumulative effect of VE and CE. Therefore, the sudden decrease of EE can be used as a criterion to judge the upper limit of AC electrode stability potential. The trend of CE and VE can be used as a reference. Activated carbon lithium-ion batteries with different lithium-ion electrolytes are charged and discharged from zer
o-charge potential constant current to various cut-off voltages of 2MA. The vertex voltage is increased from 3.4 V to 4.5 V with a step size of 0.1 V, as shown in Figure 1a. Ee, CE, and VE values as a function of vertex voltage are shown in figure 1b. According to EE criterion, the upper limit of AC electrode stability potential in EDD, EPD and PCE electrolyte is 4.0 V, 4.0 V and 4.1 V respectively, which is in accordance with our previous experimental results.
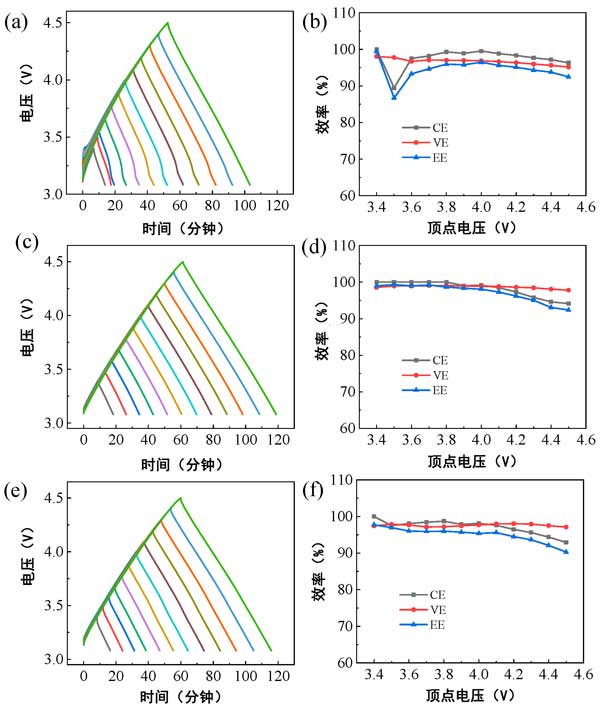
Figure 1: Constant-current charge-discharge curves from pzc to various vertex voltages in different lithium-ion electrolytes for activated carbon lithium-half cells: (a) EDD, (c) EPD, and (E) PCE; The corresponding EE, CE, and VE values are functions of the vertex voltage: (b) Edd, (d) EPD, and (F) PCE.
Overcharge to 10.0 V in addition, use the VMP3 electrochemical station to set the half-battery to charge to 10.0 V at a current of 25 Ma. The voltage and time curves of AC electrodes in EDD, EPD and Edd electrolytes are shown in Figure 2. For example, when the voltage of an EPD semi-battery reaches 5V, it drops rapidly as the charging time and capacity increase. After charging to 21.6 times rated capacity (about 4.4 hours) , the half-battery voltage drops to 4.4 V. The subsequent time is virtually unchanged, with an additional charge of 28 times the rated capacity (5.6 hours) during this period. Then, after an additional charge of 22.5 times the rated capacity (4.5 hours) , the half-battery voltage increases linearly to 6.8 V. Finally, after 14.4 hours of charging, the half-battery voltage rose sharply to 10.0 V in a vertical line, indicating that the electrolyte decomposition deposit had separated the surface of the AC cathode from the electrolyte, exposing the behavior of the dielectric capacitor. PCE activated carbon semi-battery showed similar behavior. The voltage rises to about 5V and falls to 4.6 V, then reaches a fully polarized state with less dwell time. During these processes, only very slight gases escape. In contrast, the voltage of an EDD half-cell rises to only about 8 v, with constant electrolyte breakdown and huge volume expansion. The half-battery pictures of EPD, PCE and Edd Electrolytes after overcharging are shown in figure 2b-d. For all three and a half batteries, a less noticeable temperature rise can be observed just by touching them. After the overcharge process, the voltage of the half-cell drops rapidly to 4.2-4.3 V during rest, which can be attributed to the large leakage current at voltages above the stable voltage range.
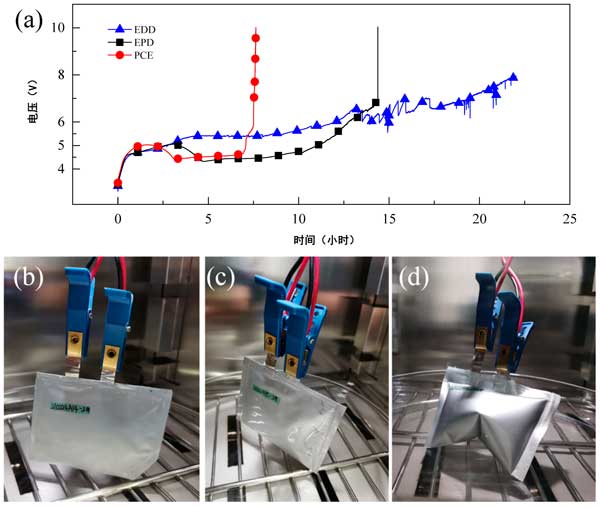
Figure 2: activated carbon lithium-ion semi-battery overcharged to 10.0 V: (a) voltage curve over time, and photographs of the latter half of the battery using different electrolytes during overcharge: (B) EPD, (C) PCE, and (D) Edd.
Since an active carbon half-cell with a PCE electrolyte can be overcharged to a high voltage of 10.0 V, it is assumed that the electrolyte-decomposed sediment separates the activated carbon electrode surface from the electrolyte through a large passivation film. At the end of the overcharge process, remove three and a half batteries from the glove box, clean the AC electrodes thoroughly with DMC solvent, and soak them in DMC overnight. The electrodes are then dried in argon. The SEM morphology and composition of activated carbon electrode surface were characterized. After the activated carbon electrode is charged to 4.5 V in PCE electrolyte, a film is formed on the surface of the electrode, which makes the edges of the activated carbon and carbon black particles not clear. After 2,000 cycles between pzc and 4.5 V, the AC electrode has been coated with a thick electrolyte decomposition deposit. By comparing the SEM images of the fresh AC electrode (Fig. 3a) and the overcharged AC electrode (Fig. 3b) , a very thick passivation film covering the surface of the overcharged AC electrode is found, which consists of two phases, that is, small spherical particles (area 1) and amorphous sediments (Area 2) . The EDS spectra showed that the content of O and S was higher in region 1, and the content of F and S was higher and the content of O was lower in region 2. The thickness of the passivation film is about 1 μm, as can be seen from the SEM cross section of Fig. 3c. Similarly, a thick passivation film is formed on the surface of the activated carbon electrode obtained by using an EPD electrolyte after overcharging of an activated carbon lithium-ion battery, as shown in Fig. 3C.

Figure 3: (a) SEM images of the alternating current electrode and the alternating current electrode from the activated carbon lithium half-cell charged to a high voltage of 10.0 V, with different electrolytes (b, C) PCE, (D) EPD.
In the overcharge performance of activated carbon in lithium-ion electrolytes, the upper stable potential limits of AC electrodes in three lithium-ion electrolytes were evaluated and determined to be 4.0-4.1 V by energy efficiency method. Then, the activated carbon lithium-ion battery was charged to 5.0 V and 10.0 V to study the decomposition of electrolyte components. Of note, self-protective passivation films are formed on the surface of the alternating current electrodes in EPD and PCE electrolytes, mainly due to insoluble breakdown products of LIFSI salts and organic solvents. It is inferred that the passivation layer consists of LIF, LI2CO3, LI2SO3, Roli, ROCO2Li and RSO2LI, and the dense passivation film is important for separating the surface of AC electrode from the electrolyte and inhibiting the further decomposition of the electrolyte. This self-protective passivation helps to prevent the lithium-ion battery from bulking up and exploding under conditions of electrical abuse and overcharging.