+86 13253371556
Activated carbon adsorption is a popular and effective physical separation method, which can be used to remove or reduce the concentration of various dissolved pollutants (organic and inorganic) in the effluent. Activated carbon has recently been found to be one of the most commonly used adsorbents and can be used effectively to remove a variety of pollutants from air, soil and liquids. This development is very noteworthy. Most adsorption occurs on the pore walls of particles, which is why adsorbents are often classified as porous solids. The presence of pollutants in water is a very important environmental problem that needs to be solved. This work describes the development and testing of new types of activated carbon that can effectively remove methyl red from aqueous solutions, thus providing a solution to this problem.
the objective of the experiment is to study the use of orange peel activated carbon as an adsorbent for methyl red adsorption. Batch adsorption studies (10-60 minutes) and pH (3-11) were carried out by monitoring various basic parameters, such as adsorbent dose (0.25-1.25 g/L) , methyl red concentration (100-400 mg/L) , temperature (40-60 ° C) , contact time. The batch reactor is where all the adsorption research is done. Before and after adsorption, the concentration of methyl red dye in the supernatant was measured by uv-vis photometer. The concentrations in the effluents were then calculated with the help of a standard calibration curve. Each experiment was performed several times in a controlled setting, and the average value was used for subsequent calculations. Fig. 1 shows the experimental setup for bulk adsorption studies.

Characterization of a new type of activated carbon
Activated carbon is the adsorbent used in this experiment. The physical chemistry properties (typical properties, approximate and final analysis) of the new activated carbon were used to determine its characterization. SEM is used to examine the surface morphology of activated carbon samples. XRD was performed by X-ray diffractometer. Functional groups were identified by recording FTIR spectra in the range of 4,000-500 CM-1. Analysis was performed using Brunauer-Emmett-Teller (Bet) . BET surface area, pore volume and pore diameter of activated carbon were measured by nitrogen adsorption method. Thermogravimetric or“Dry weight loss” methods are usually used to determine moisture content. In this method, the sample is heated and the weight loss due to water evaporation is measured. The combination of a drying chamber and a balance, as well as a moisture analyzer, is a common moisture analysis technique. Weigh a small amount of activated carbon and transfer it to a glass plate. It is then heated in the oven at a temperature above 100 ° C for 2 hours. Remove the pan from the oven and cool. The weight of the dried sample was determined after cooling. According to the equation used to calculate the moisture content (weight percentage) .
The content of volatile matter was measured directly in automatic approximate analyzer. When the fuel oil sample is burned completely, the non-combustible residue is reflected by the ash content. High temperature muffle furnaces capable of withstanding temperatures between 300 and 400 ° C. In the presence of oxygen in the air, some substances (such as water and other volatile elements) will burn, while others (including organic molecules) will evaporate. The ash content can be calculated by comparing the weight of the samples before and after ashing. Fixed carbon is calculated by subtracting the percentage of volatile matter, moisture content and ash content from 100. The bulk density of activated carbon is by means of a densimeter. Energy dispersive X-ray analysis of the chemical composition of activated Carbon Fig. 2. EDAX analysis of activated carbon has C, O, Zn and other elements.
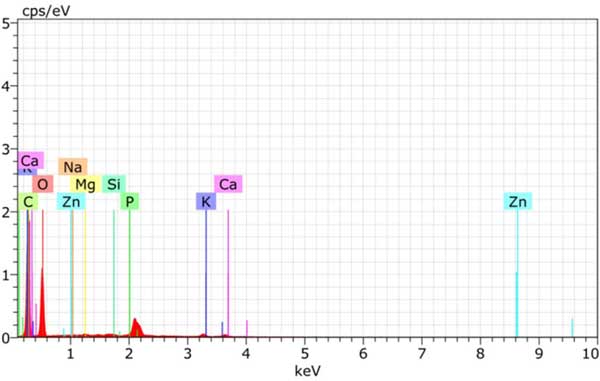
Activated Carbon Edax
Pore structure of activated carbon obtained by Scanning electron microscope analysis. SEM equipment with ultra-high resolution low voltage imaging and unique low vacuum capability. SEM images were taken at 15kV with different resolutions of 1nm, 1.4 nm at 1kV, 1.8 nm and 30PA at 3KV. The surface physical morphology of activated carbon is observed by means of Sem. Fig. 3 shows the SEM image of activated carbon. This shows that the outer surface of activated carbon has a uniform open macropores. In addition, the surface of activated carbon is highly porous and characterized by heterogeneity. It is also clear that the activated carbon has micropores and cavities on its outer surface, where the dark zone is located. After carbonization, the active carbon forms voids (called pores) due to evaporation of ZNCL2. These holes can be thought of as channels to a network of micropores.

SEM image of activated carbon.
Batch adsorption of methyl red by activated carbon
Dye adsorption on activated carbon was tested under various conditions to see how initial solution concentration affected overall adsorption time. The study was carried out at 60 ° C and ph 7 with a constant adsorbent dose of 1 g/L and an initial methyl red concentration ranging from 100 to 400 mg/L. The absorption of dyes by activated carbon is rapid in the first 40 minutes, then occurs at a slower rate and eventually reaches saturation. When the initial methyl red concentration increased, the equilibrium dye removal rate decreased from 100 mg/l to 400 mg/L. At low concentrations, almost all methyl macromolecules are adsorbed on the surface, and the high amount leads to the fast saturation of methyl red surface.
The adsorption capacity of a given initial adsorbent concentration depends on the dosage of the adsorbent. Therefore, this parameter is critical. The removal efficiency of methyl red by activated carbon with different adsorbent dosages was studied. The amount of adsorbent was increased by 1 g/L as a percentage of removal and remained almost the same after that. An increase in the adsorption dose results in an increase in the available surface area, which increases the availability of additional adsorption sites. Further increase leads to saturation of the adsorption site. Therefore, it goes without saying that equilibrium is achieved faster at lower concentrations.
Comparative study, after batch adsorption test data, the results of the new activated carbon and the local available commercial activated carbon were compared.
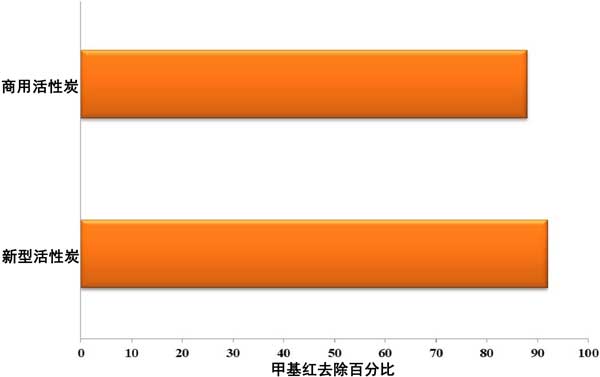
Comparative experimental data of activated carbon
The comparative data of new type activated carbon and activated carbon at 60 ° C, 11 ph, 100 mg/L adsorbent dosage is 1 g/L. The concentration of methyl red, the amount of adsorbent used, pH value and adsorption temperature are the main variables which affect the removal efficiency of methyl red. It was observed that the adsorption efficiency increased with the increase of solution temperature (60 ° C) , contact time (60 minutes) and adsorbent dose (1 g/L) , but decreased with the increase of initial dye concentration. The increase in pH results in more methyl red removal, which is optimal at a pH of 11. The isotherms of Langmuir and Freundlich are used to fit the equilibrium data. Langmuir provides a better fit, at 60 ° C it is 111.11 mg/g. The comparison of the new activated carbon shows that the removal percentage of the new activated carbon is higher than that of the ordinary activated carbon.