+86 17737144966
Refining is an essential process in petroleum processing, which uses a large number of chemical products and produces solid waste and wastewater, which are considered hazardous waste. In fact, refinery wastewater contains a lot of heavy metals, phenols, benzene and COD. In recent years, refinery wastewater pollution has increased due to the increased use of petroleum products by industry and cities. Treatment of refinery wastewater by adsorption was chosen on the basis of its high potential for the removal of toxic compounds and its cost-effectiveness. This issue shows how activated carbon can be used to effectively remove sulfur pollutants from refinery wastewater.
By calcination and activation with HNO3, H2O2 and KOH, the waste cotton fibers were successfully transformed into activated carbon with different structures. Figure 1 shows the SEM images of the three activated carbons. It can be seen that chemical activation contributes to the formation of macropores. A hollow core with a diameter of 0.5-5.0 μm can be observed in the whole length of the fiber. Activated carbon has a highly porous structure with a core diameter between approximately 0.5 and 5 μm (Fig. 1b, D) . This macroporosity may be related to the chemical destruction of lignin in fiber inner wall. The reference activated carbon (Fig. 1a) is macroporous and characterized by the presence of a fiber core whose diameter depends on the activator. The largest diameter was obtained using hydrogen peroxide as the activation reagent (figure 1B) . As shown in figure 1b-d, the precursor structure (initially with a clean surface) is destroyed after each chemical treatment. Interestingly, the surface of one of the activated carbons changed significantly under the influence of chemical activation (compared with powdered activated carbons,granule activated carbon) .
1: SEM image of activated carbon: (a) reference activated carbon, (B) HNO3 activation, (C) H2O2 activation, and (D) Koh activation.

Bed adsorption experiment
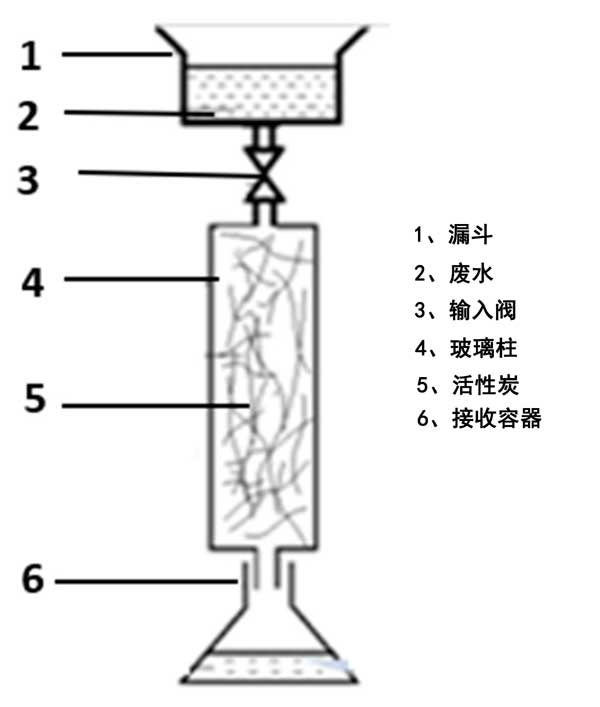
A batch adsorption equilibrium experiment was carried out by reacting a known amount (4.0 g) of activated carbon with 50 ml of actual wastewater sample in a sealed glass bottle. Five samples with different initial sulfur concentrations (100,300,500,800 and 1000 mg · L -1) were tested. Place the bottle on a thermostat shaker for 24 hours until balanced (final pH: 10.5) . A schematic diagram of the apparatus for the bed adsorption experiment is shown in Fig. 2. Carbon fibers were packed into glass columns to study their adsorption capacity. The wastewater is fed into a funnel and passed through an input valve to regulate the flow rate to 4-5.10 m3 · H-1. First, the wetting characteristics of activated carbon fibers are improved by using a column-flow wetting bed adsorption device with distilled water. Second, the contents of the refinery wastewater sample in the funnel are allowed to be discharged to the receiving container by gravity through the activated carbon bed. Recycle treated water every hour.
Fig. 2: Bed Adsorption Experiment.
Fixed bed adsorption results
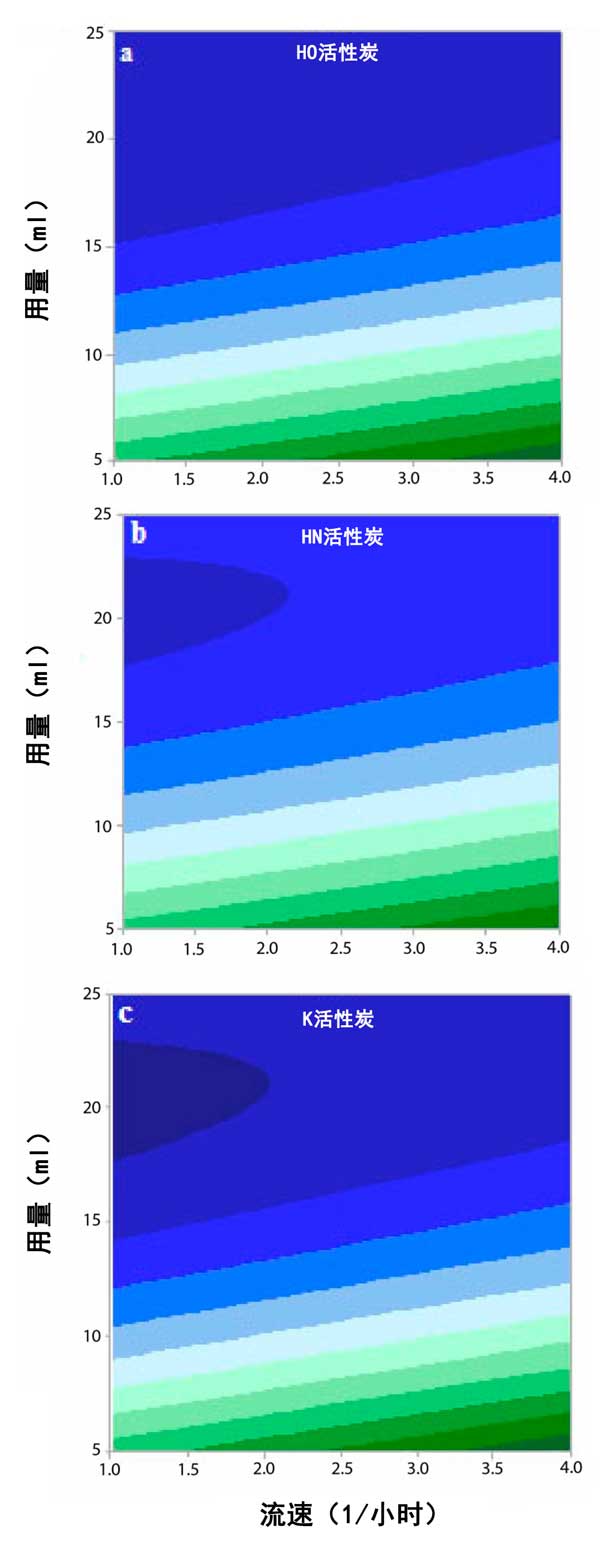
An adsorption bed column is installed as shown in Figure 3 to allow the refinery wastewater to flow under gravity. The diameter and height of the bedposts were 5.4 cm and 90 cm, respectively. The optimum adsorption conditions, including the ratio of activated carbon and the flow rate of the efflux, were determined according to the experimental parameters. Response surface method was used to design the experiment. The results were analyzed by analysis of variance to check the effect of parameters on COD of treated wastewater. For each of the 16 experiments, calculate COD. Figure 3 shows the effect of activated carbon and wastewater flow rate on COD in the reclaimed wastewater. For all activated carbon samples with high wastewater flow rate, COD was increased by increasing the dose quality. However, at low flow rates, COD decreases. In a word, OH activated carbon is the most effective adsorbent (compared with HN activated carbon and K activated carbon) for reducing COD of refinery wastewater.
Figure 3: isograms of COD, (a) OH, (b) HN and (c) K activated carbons.
Sorption isotherm modeling
The equilibrium Sorption isotherm of sulfur compounds were modeled and studied on HO activated carbon from aqueous solutions. To describe the Sorption isotherm data, five mathematical models were used. The main objective of this work is to select the most appropriate model to describe the experimental results of the Sorption isotherm, to determine the theoretical Sorption isotherm, and to give the parameters. The initial goal was to develop a two-parameter equation to simulate the adsorption of sulfur compounds by HOAC. The experimental conditions were as follows: the initial sulfur concentration ranged from 5 to 300 mg · L -1, stirring at 150 rpm, and a controlled temperature of 25 °C. For all isothermal models, the experimental values of QE and CE are used to study the linear equations to determine their linear parameters. The linear correlation coefficient (R 2) was used to evaluate the correlation between theoretical and experimental values. We can conclude that the isotherm parameters are different. Langmuir model has the highest correlation coefficient (R2 = 0.98) . This indicates that the adsorption of sulfur compounds on synthetic activated carbon follows a Langmuir equation. However, the monolayer adsorption capacity obtained using the Langmuir isotherm is higher than that obtained using other isotherm models. This shows that all the active sites of synthetic activated carbon are similar and there is no interaction between sulfur compounds.
The application of activated carbon in the removal of sulfur compounds from oil refinery was studied. An environmentally friendly activated carbon was successfully synthesized as an adsorbent for sulfur compounds in refinery wastewater. The kinetic and equilibrium data of sulfur compounds were obtained, and the Langmuir and pseudo-second order models were fitted respectively. The results showed that the pore size distribution, pore volume and surface area of activated carbon were successfully increased by adding hydrogen peroxide, nitric acid and potassium hydroxide. In addition, compared with HN activated carbon and K activated carbon, HO activated carbon has the best ability to remove sulfur compounds. According to the analysis of response and variance, it is found that the efflux velocity is the most effective factor to reduce COD of refinery wastewater in the fixed-bed adsorption process. The Langmuir model shows the best fit for sulfur compound adsorption (R2 = 0.98) , which means that their adsorption on synthetic activated carbon is homogeneous. The pseudo-first order, pseudo-second order and intra-particle diffusion equations were used to test the kinetic data. The pseudo-second order equation shows the best description of the kinetic data (R2 = 0.99) , which means that this adsorption can be limited by the chemical adsorption process.